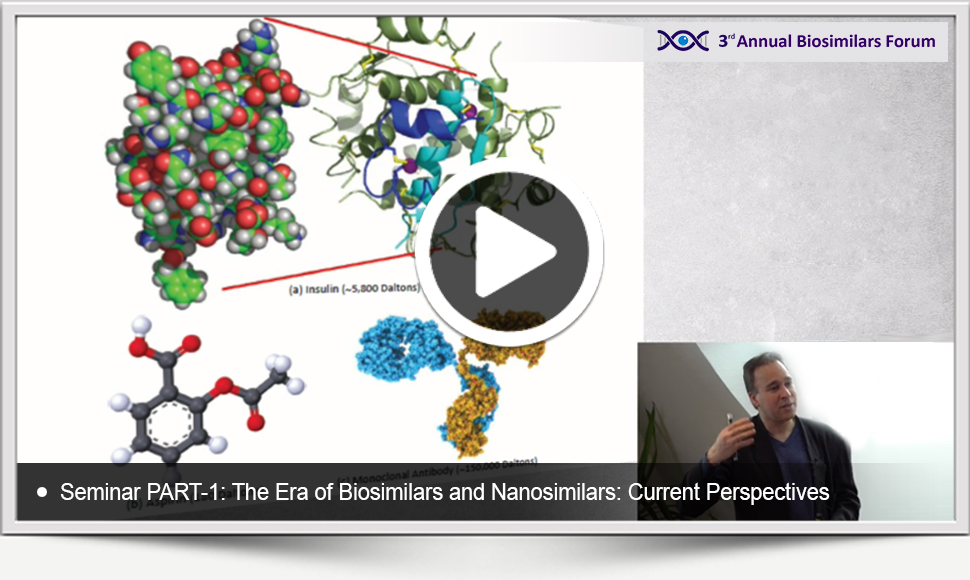
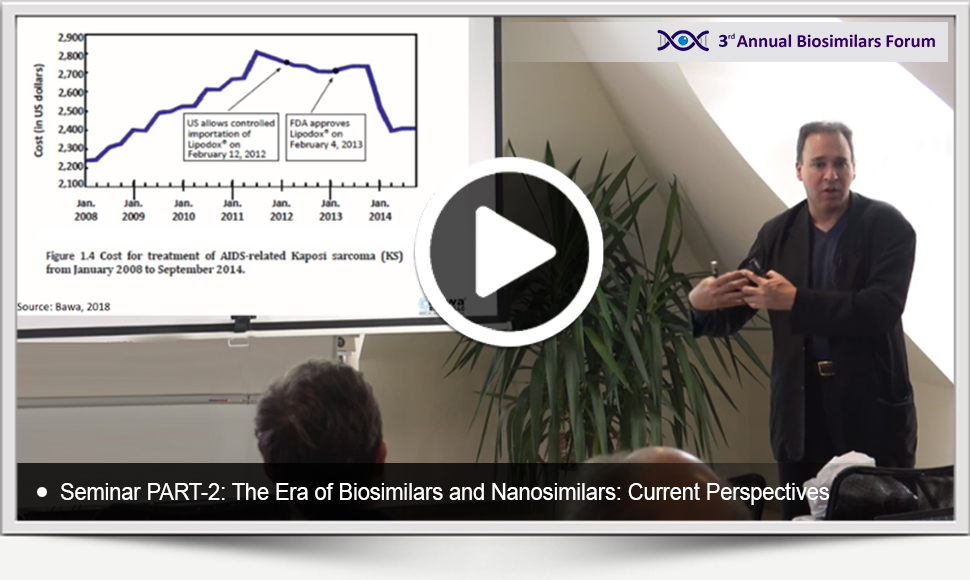
The presentation materials of the Seminar entitled The Era of Biosimilars and Nanosimilars: Current Perspectives delivered by Raj BAWA at the 3rd Annual Bioaimilars Forum on the 27th of October, 2018 in Budapest are available below to watch and download as a courtesy of the event series. Here are the links to reach the videos and your copies of the materials:
Presentation videos of ‘The Era of Biosimilars and Nanosimilars: Current Perspectives’ Seminar
|
President of Bawa Biotech LLC | Adjunct Professor at Rensselaer Polytechnic Institute, USA | Scientific Advisor at Teva Pharmaceutical Industries, Israel | Founding Director of American Society for Nanomedicine | Learn more… |
Downloadable presentation materials
Presentation material of the Seminar: The Era of Biosimilars and Nanosimilars: Current Perspectives (.PDF, 30 674 KB)
CBS News Video: What is Nanotechnology? (Online Video File)
… and the author’s special gift to you:
Downloadable book chapter: Current Immune Aspects of Biologics and Nanodrugs: An Overview (.PDF, 7743 KB)
Book overview: Pan Stanford Series on Nanomedicine Vol. 3: Immune Aspects of Biopharmaceuticals and Nanomedicines (.PDF, 3878 KB)
Discover more about the filed of nanomedicine! Please visit the website of the American Society for Nanomedicine for related relevant information and more free downloads: Resource Center
Feel free to ask  ‘ experts!
‘ experts!
Do you need more professional help?
We hope our professional team at Biosimilars Forum series and at Accelsiors CRO can contribute to your success by sharing these valuable materials and that we can support your work efficiently in the complex field of innovative drug development.
If you need more support or professional help in connection with any aspect of clinical drug research and development, or you are interested in receiving more information about our events, do not hesitate to contact Accelsiors’ Clinical Development Team by using this next form!
Let us to accelerate your drug development program by sharing with you our expertise and well-tailored global service capabilities! Learn more about Accelsiors CRO!
About the Author
 Raj Bawa, MS, PhD, is president of Bawa Biotech LLC, a biotech/pharma consultancy and patent law firm based in Ashburn, Virginia. He is an inventor, entrepreneur, professor and registered patent agent licensed to practice before the U.S. Patent & Trademark Office. Trained as a biochemist and microbiologist, he has extensive expertise in pharmaceutical sciences, biotechnology, nanomedicine, drug delivery, microbial biodefense, FDA regulatory issues, and patent law. Since 1999, he has held various positions at Rensselaer Polytechnic Institute in Troy, NY, where he is an adjunct professor and where he received his doctoral degree (biophysics/biochemistry). Currently, he serves as a scientific advisor to Teva Pharma, Israel, is a visiting research scholar at the Pharmaceutical Research Institute of Albany College of Pharmacy in Albany, NY, and is vice president of Guanine, Inc., based in Rensselaer, NY. He has served as a principal investigator of SBIRs and reviewer for both the NIH and NSF. Currently, he is principal investigator of a CDC SBIR Phase 1 grant to develop an assay for carbapenemase-resistant bacteria. In the 1990s, Dr. Bawa held various positions at the US Patent & Trademark Office, including primary examiner from 1996–2002. He is a life member of Sigma Xi, co-chair of the nanotech and personalized medicine committees of the American Bar Association and founding director of the American Society for Nanomedicine. He has authored over 100 Key Award from Rensselaer’s Office of Alumni Relations (2005); and Lifetime Achievement Award from the American Society for Nanomedicine (2014).
Raj Bawa, MS, PhD, is president of Bawa Biotech LLC, a biotech/pharma consultancy and patent law firm based in Ashburn, Virginia. He is an inventor, entrepreneur, professor and registered patent agent licensed to practice before the U.S. Patent & Trademark Office. Trained as a biochemist and microbiologist, he has extensive expertise in pharmaceutical sciences, biotechnology, nanomedicine, drug delivery, microbial biodefense, FDA regulatory issues, and patent law. Since 1999, he has held various positions at Rensselaer Polytechnic Institute in Troy, NY, where he is an adjunct professor and where he received his doctoral degree (biophysics/biochemistry). Currently, he serves as a scientific advisor to Teva Pharma, Israel, is a visiting research scholar at the Pharmaceutical Research Institute of Albany College of Pharmacy in Albany, NY, and is vice president of Guanine, Inc., based in Rensselaer, NY. He has served as a principal investigator of SBIRs and reviewer for both the NIH and NSF. Currently, he is principal investigator of a CDC SBIR Phase 1 grant to develop an assay for carbapenemase-resistant bacteria. In the 1990s, Dr. Bawa held various positions at the US Patent & Trademark Office, including primary examiner from 1996–2002. He is a life member of Sigma Xi, co-chair of the nanotech and personalized medicine committees of the American Bar Association and founding director of the American Society for Nanomedicine. He has authored over 100 Key Award from Rensselaer’s Office of Alumni Relations (2005); and Lifetime Achievement Award from the American Society for Nanomedicine (2014).
Copyrights notice
© Raj Bawa. All rights reserved. All of these presentation materials and articles and the content of these are owned by the author of the materials and are protected by copyright laws and international treaty provisions. Reproduction of any material on these lectures and articles is only authorized for exclusive information for strictly personal and private use. Any reproduction and/or representation of all or part of the lectures and articles, or any of the elements that compose it, on any media whatsoever, for any other purpose, including commercial, is expressly prohibited, unless the prior written authorization from the author.